
Phosphorylated full‐length Tau interacts with 14‐3‐3 proteins via two short phosphorylated sequences, each occupying a binding groove of 14‐3‐3 dimer - Neves - 2021 - The FEBS Journal - Wiley Online Library
Phosphorylated full-length Tau interacts with 14-3-3 proteins via two short phosphorylated sequences, each occupying a binding g
IJMS | Free Full-Text | The 14-3-3 Proteins as Important Allosteric Regulators of Protein Kinases | HTML

14-3-3 protein directly interacts with the kinase domain of calcium/calmodulin-dependent protein kinase kinase (CaMKK2) - ScienceDirect

An example of least-squares parameter estimation exhibiting multiple... | Download Scientific Diagram

14-3-3 prevents dephosphorylation of Thr 108 by protein phosphatase 2A.... | Download Scientific Diagram

Glaciokarst features of Mt. Tymphi: Schichttreppenkarst (A) as well as... | Download Scientific Diagram

A proposed model for potassium-selectivity in CPA1s. Side view (a, b),... | Download Scientific Diagram
Biomolecules | Free Full-Text | Cation Transporters of Candida albicans—New Targets to Fight Candidiasis? | HTML
Phosphorylated full-length Tau interacts with 14-3-3 proteins via two short phosphorylated sequences, each occupying a binding g

Spherical concretions. A. Spherical boulder; B. Inner structure; C.... | Download Scientific Diagram

Crystal structure of the complex USP8pS718/14-3-3f. (A) Assembly of the... | Download Scientific Diagram

Undersaturation index Eq. (33) as a function of inverse flow rate 1/Q.... | Download Scientific Diagram

The CPA motif is located in the core domain, near the ion binding site.... | Download Scientific Diagram

Phosphorylated full‐length Tau interacts with 14‐3‐3 proteins via two short phosphorylated sequences, each occupying a binding groove of 14‐3‐3 dimer - Neves - 2021 - The FEBS Journal - Wiley Online Library

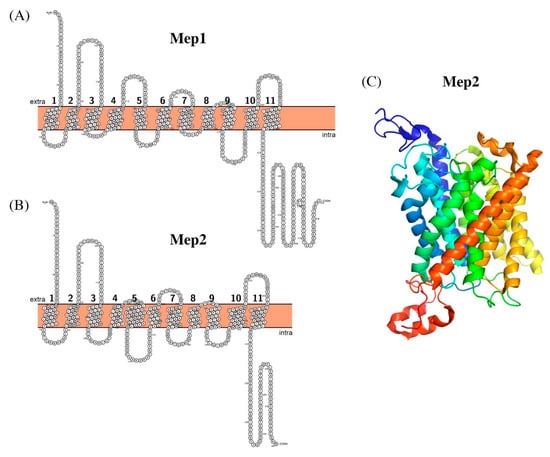
Sequence analysis of YNL321W. A, topological model based on predictions... | Download Scientific Diagram

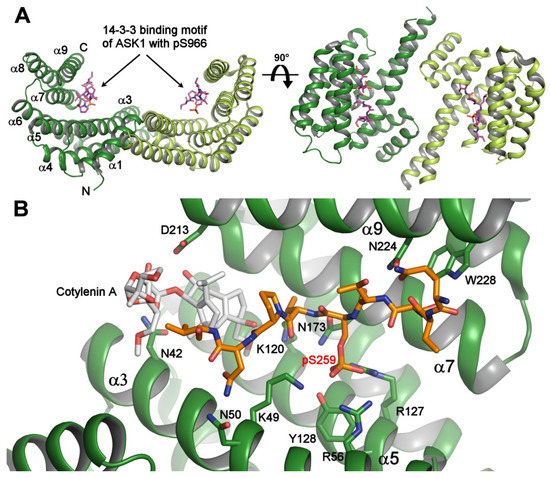
Antioxidants | Free Full-Text | Prdx1 Interacts with ASK1 upon Exposure to H2O2 and Independently of a Scaffolding Protein | HTML

Phosphorylated full‐length Tau interacts with 14‐3‐3 proteins via two short phosphorylated sequences, each occupying a binding groove of 14‐3‐3 dimer - Neves - 2021 - The FEBS Journal - Wiley Online Library

Phosphorylated full‐length Tau interacts with 14‐3‐3 proteins via two short phosphorylated sequences, each occupying a binding groove of 14‐3‐3 dimer - Neves - 2021 - The FEBS Journal - Wiley Online Library





